Equations > Chemistry > Thermochemistry > Gibb's Free Energy Change in Terms of Number of Moles, Faraday, and Standard Reduction Potential
Gibb's Free Energy Change in Terms of Number of Moles, Faraday, and Standard Reduction Potential
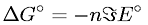
Latex Code:
MathML Code:
MathType 5.0:
