Equations > Chemistry > Thermochemistry > Gibb's Free Energy Change in Terms of Gas Constant, Absolute Temperature, and Equilibrium Constant
Gibb's Free Energy Change in Terms of Gas Constant, Absolute Temperature, and Equilibrium Constant
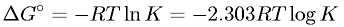
Latex Code:
MathML Code:
MathType 5.0:
